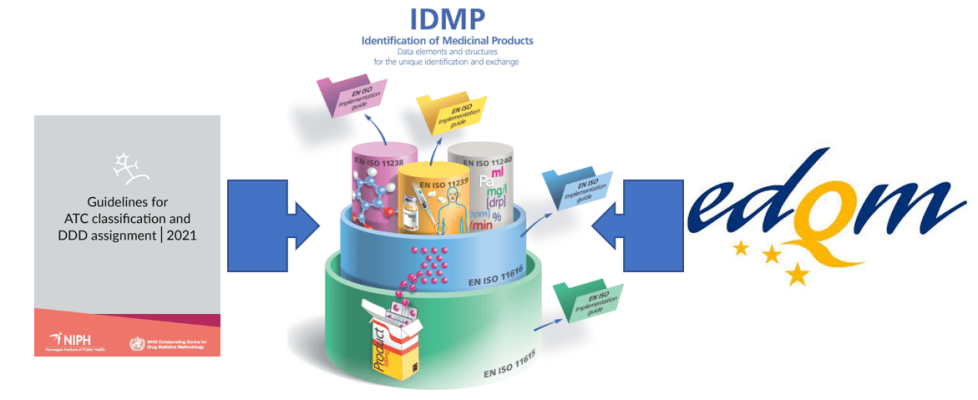
The importance of medicinal product dictionaries (MPDs) is highlighted by two concrete examples. MPDs bring together the information on medicinal products validated by a National Competent Authority and a variety of sources. Making sure that the identification of the medicinal product across these sources can be done automatically and without error, makes the implementation of IDMP such a crucial task. In addition to the IDMP standards, and as an example of supplementary sources of data, we will also highlight the possible use of the Melclass database on the classification of medicines and their supply conditions (maintained by EDQM), as well as the ATC (and DDD) classification as maintained by the WHO-CC for Drugs Statistics Methodology.!
Date: Thursday 29 April 2021
Time: 3:00 – 4:30 pm CEST
Presenters:
Leonora Grandia – Z-Index
Ursula Tschorn – Pharmazie.com
François-Xavier Lery – EDQM
Christian Lie Berg – WHO-CC for Drugs Statistics Methodology
Markus Pongratz – AGES -Melclass Expert
Moderators:
Christian Hay (GS1 and ISO TC 215/WG6 – WP 1)
Robert Stegwee (CEN TC 251 – WP 1)
You can already register with the following link:
https://us02web.zoom.us/webinar/register/WN_9NTL4DyaQBCgruwV_NAdRg