
- This event has passed.
Community of Expertise: IDMP data : from source to final use
Description:
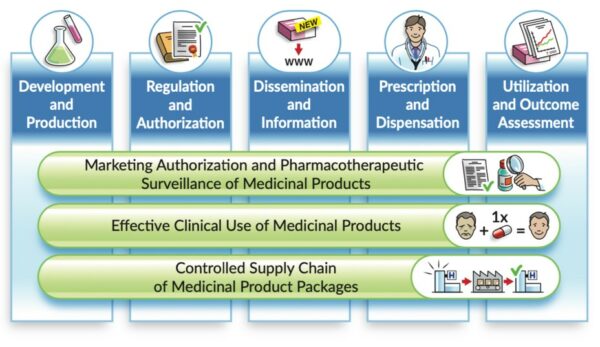
Referring to the landscape of medicinal product data – from “research and development” to “utilisation and outcome assessments”- the vision is that the essential IDMP identifiers remain used and unchanged along that life cycle.
This Community of Expertise (CoE) will provide opportunity :
for industry to present their perspective and needs, with large international companies, with SME facing IT investments to implement IDMP, and with advisers supporting IDMP implementation in companies and organisations;
for Medicinal Product Dictionaries suppliers, with their the hub function between pharmaceutical companies, National Competent Authorities, on one hand, and for their customers in the clinical space on the other hand;
and for the consumption of these data with Pharmacovigilance.
Presenters:
Quentin Darrasse, Roche Pharma
Andreas Franken, BAH
Stefan Peev, NNIT
Frederic Doc, Vidal
Boukje Raemaekers, WHO-UMC
Panellist:
Malin Fladvad, WHO-UMC
Moderators:
Christian Hay, UNICOM WP1, ISO/TC 215 WG6 Pharmacy and Medicines Business, GS1
Robert Stegwee, UNICOM WP1, CEN TC 251
